Haleon Consumer Healthcare Canada is voluntarily recalling two lots of Advil Cold and Sinus Daytime/ Nighttime Convenience Pack (18 and 36 caplets). This recall is for retail stores and wholesalers only and is due to incorrect labelling.
This recall only impacts two lots of Advil Cold and Sinus Daytime/ Nighttime Convenience Pack 18ct and 36ct, lots ER2069 and ER2072 and does not impact any other Advil Cold & Sinus products.
This voluntary recall is being conducted in coordination with Health Canada. It has been classified as “Type II”, meaning that the use of, or exposure to, the impacted product may cause temporary adverse health consequences, and has a remote probability of causing serious adverse health consequences.
Consumers with Advil Cold and Sinus Daytime/ Nighttime Convenience Pack 18ct and 36ct, lots ER2069 and ER2072 should stop use of the product and contact our Consumer Relations team via email at mystory.ca@haleon.com, or by phone at 1-888-788-8181 Monday – Friday, 08.00 - 18.00 EST
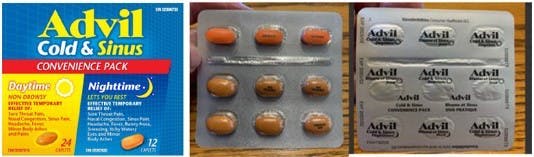
Pictured above:
Left –Convenience pack carton with photos for the Daytime and Nighttime caplets.
Center – Appearance of caplets in blister card. Note 3 Nighttime (orange) caplets on the top row and 6 Daytime (beige) caplets on the bottom row.
Right – Example mislabeled blister card back


